
Now, balance the reaction using fractions such that we get one mole of product. 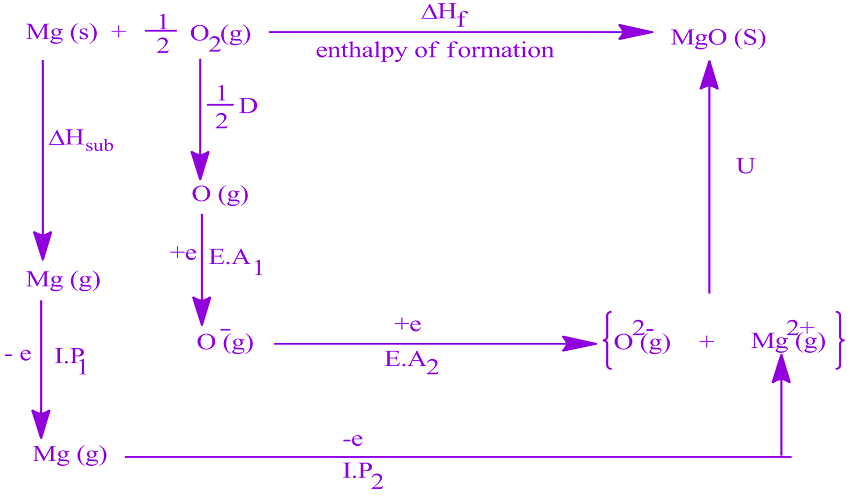
Step 1: Write the overall reaction of the formation of Lithium Fluoride from the individual elements. Let’s understand how to use Born Haber cycle by drawing the cycle of Lithium Fluoride and write down the Hess’ Law for it. Now let’s try to understand how to use a Born-Haber process by an example. Hess Law of constant heat summation states that for a multi-stage reaction with various intermediate reactions, the enthalpy change for the overall reaction is the sum of the enthalpy changes of the intermediate reactions. The cycle involves the use of Hess’ law extensively to find this lattice energy. This energy is due to the electrostatic force of attraction. Lattice Energy is the energy released when free anions and cations come closer to form a lattice structure. The Born Haber cycle is mainly used to calculate the lattice enthalpy of various ionic compounds, otherwise, it is difficult to find. A Polish-American Chemist, Kazimierz Fajans, developed the same cycle independently. The cycle was developed by contribution from both of them in the year 1919. Where all the numbers you plug in are positive.The Born-Haber process is named after the two famous German Scientists Max Born and Fritz Haber. #color(blue)(DeltaH_"lattice" -= -|DeltaH_"lattice"|)# Solving for #DeltaH_"lattice"# usually gives a positive answer, so we take the negative of the answer by convention to get: Take the step as being upwards to generate a complete cycle, for which #DeltaH_"cycle" = 0# (since #H_f = H_i# for a complete cycle). #"These cancel out completely upon adding, proving"#Īnd now if we wish, the lattice energy can be calculated. Put this all together, with some data, and we get, for #"1 mol"# of #"NaCl"(s)#: #"Cl"(g) + e^(-) -> "Cl"^(-)(g)#lArr# chlorine was a gas, and now needs to gain an electron, the definition of electron affinity. 
#1/2"Cl"_2(g) -> "Cl"(g)#lArr# chlorine is now made atomic (defines bond energy).#"Na"(g) -> "Na"^(+)(g) + e^(-)#lArr# Ionization of the gas to remove an electron is by definition the ionization energy.#"Na"(s) -> "Na"(g)#lArr# sublimation of sodium solid.Our goal is to transform the reactants into their ionic gases, as that is the reaction that describes the process for which "lattice energy" is defined. We begin by writing the formation reaction, which is by definition from the elemental states at "C"# and #"1 atm"#: The Born-Haber cycle takes advantage of the state function property of the change in enthalpy to indirectly determine the lattice energy of ionic compounds through processes that utilize known thermodynamic quantities like ionization energy and electron affinity. By definition, the gaseous cation and anion forming the corresponding ionic compound release energy termed the lattice energy, the energy contained within the lattice structure.įor an alternative explanation, see here.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
